T1D Guide
T1D Strong News
Personal Stories
Resources
T1D Misdiagnosis
T1D Early Detection
Research/Clinical Trials
Why the ISLET Cell Act Matters Now for People With Type 1 Diabetes
The ISLET Cell Act could expand access to islet cell transplantation and accelerate the development of new cell-based therapies for people living with type 1 diabetes (T1D). Learn why this important legislation matters now and how it could shape the future of T1D treatment, and most importantly, lead the path to a cure.

In the last 10 years, disease-modifying therapies (DMTs) have progressed rapidly in the diabetes research field, with therapies that delay the onset of T1D (Tzield), therapies that sustain beta cell function in stage 3 T1D (Diamyd) and tolerogenic medication designed to retrain the immune system in newly diagnosed T1Ds (COUR Pharma).
And while these therapies are incredible for the path forward, they aren’t an out-and-out cure.
Other entities leveling the playing field are stem cell-manufactured cells, which can be transplanted into a T1D patient to enable the person to produce their own insulin. As type 1 diabetes is an autoimmune disease in which the body mistakenly attacks its insulin-producing cells, scientists are working on encapsulation devices to protect these cells (Sernova) and newer autoimmune drugs that don’t have the dangerous side effects of the past (tegoprubart).
Breakthrough T1D (Formerly JDRF) recently reported its stance on the ISLET Act and its correlation to cell therapy funding worldwide. “We are currently funding over $450 million in cure research in 23 countries, which includes the University of Chicago’s exciting trial testing Eledon’s tegoprubart. While it takes time to ensure medical breakthroughs like cell therapies are safe and accessible to everyone in our community, we are leading the charge for swift and safe access to these potentially life-changing treatments.”
But for these great strides to come to fruition, one very important bill needs to be corroborated, the ISLET Cell Act, which is gaining attention now following recent life-saving transplant surgeries that have left type 1 patients insulin-independent.

The legislation could accelerate access to islet cell transplantation and cell-based therapies, offering new hope for people living with T1D.
What Is the ISLET Cell Act?
The Increase Support for Life-saving Endocrine Transplantation (ISLET) Act is proposed U.S. legislation (H.R.4304/S.3105) that reclassifies transplanted pancreatic islet cells from a “biological drug” to an “organ.” By removing them from FDA drug regulation, the bill aims to improve access to islet transplantation for people with type 1 diabetes and to align U.S. standards with international practices.
As a result, this regulatory shift would amend the Public Health Service Act to define human cadaveric islets as organs, allowing them to be regulated by the Health Resources and Services Administration (HRSA) rather than as drugs by the FDA.
The bill’s purpose aims to reduce regulatory hurdles, improve patient access to treatment, and boost innovation in islet transplantation for T1D patients.
As of late 2025-early 2026, the legislation has been reintroduced, with support from researchers such as Dr. Camillo Ricordi and organizations like the Diabetes Research Institute (DRI) Foundation, and Breakthrough T1D, who recently submitted a proposal to the U.S. Department of Health and Human Services (HHS) requesting that HHS reclassify deceased donor pancreatic islets as organs.
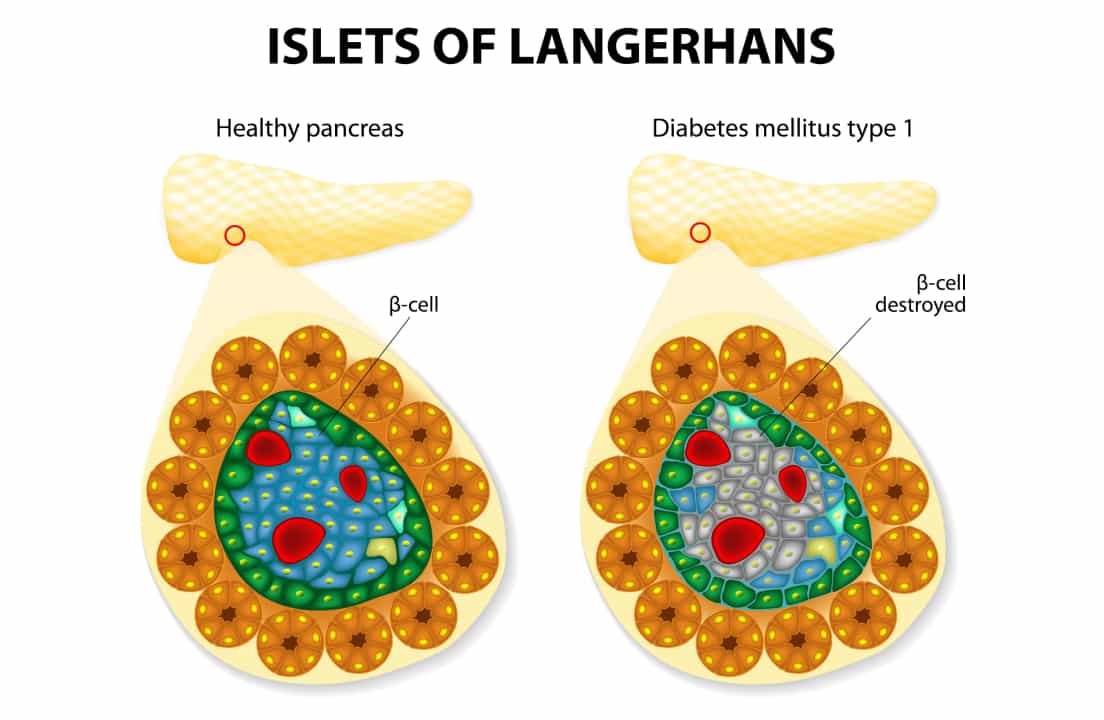
What Are Islet Cells and Why Are They Important in Type 1 Diabetes?
Islet cells (Islets of Langerhans) are small clusters of cells scattered throughout the pancreas that detect blood sugar levels and release hormones, primarily insulin from beta cells, to maintain normal glucose levels.
The destruction of beta cells leads to T1D, as the immune system mistakenly attacks these insulin-producing cells, resulting in severe insulin deficiency and high blood sugar.
Why the ISLET Cell Act Matters Right Now
The ISLET Cell Act is gaining momentum now as advances in transplantation, particularly at the University of Chicago, where Dr. Piotr Witkowski has completed several islet cell transplantation surgeries using cadaver islets, followed by the immunosuppressant drug tegoprubart.
These recent advances in cell therapies have renewed urgency around improving access to islet cell treatments. However, current regulatory barriers have slowed access. The current FDA regulatory structure treats islet-like drugs, creating challenges for hospitals and transplant centers.
Additionally, the transplant surgery is only available to type 1 individuals who experience severe hypoglycemia and unstable glucose episodes that impair their daily lives.

The ISLET Cell Act could Improve Access to Islet Cell Transplants.
If this legislation passes, it could allow more transplant centers to offer islet cell procedures and support innovation.
It’s important to note that the islet cell legislation is not a cure and limitations still exist, including donor supply and the need for immunosuppressive therapy.
Significantly, the ISLET Cell Act could support the development of next-generation treatments, including stem-cell–derived islets and other regenerative approaches.
How the Islet Cell Act Could Help People With Type 1 Diabetes
- Expand the availability of islet transplantation programs.
- Encourage research and innovation.
- Potential to improve the quality of life for people with brittle diabetes.
- Bridge toward future functional cures.
Limitations and What the Act Does Not Do
- It is not a cure for T1D.
- Transplants still require immune suppression.
- There is now a limited donor availability.
- Ongoing need for new technologies.
What This Means for the Future of Type 1 Diabetes Treatment
There’s no doubt, the ISLET Act’s role in advancing cell replacement therapies is monumental. With its potential to combine stem-cell research and immune therapies, it could be life-changing for many who struggle with dangerously unstable glycemic control.
The future of this bill has many advocates and researchers watching closely to see how these developments may shift the focus of research and technology advancements. Speak to your representatives and learn more about what you can do to support the ISLET Act.
In conclusion, the Islet Cell Act is an important step. It enhances the growing momentum toward restoring insulin production internally.


.webp)





.webp)


.jpg)







.jpeg)


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)



.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)

.jpg)

.jpg)

.jpg)
.jpg)



.jpg)



.jpg)




.jpg)
.jpg)

.jpg)
.jpg)


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)



.jpg)
.jpg)
.jpg)

.jpg)

.jpg)














.jpg)


.jpg)







.webp)







.webp)