T1D Guide
T1D Strong News
Personal Stories
Resources
T1D Misdiagnosis
T1D Early Detection
Research/Clinical Trials
Type 1 Diabetes Breakthroughs to Watch in 2026
2026 brings major breakthroughs for people living with type 1 diabetes (T1D). Here, we highlight new discoveries focusing on stem cell-derived islet replacements, targeted immunosuppression therapies, encapsulation technology and hypoimmune therapies in the works.

These key advancements include the anticipated approval of Vertex’s stem cell therapy, the use of tegoprubart to eliminate the harmful effects of past immunosuppressive medications in transplant surgeries, and new gene-edited hypoimmune islet cells developed by Sana Biotechnology.
With updates from the Advanced Technologies & Treatments for Diabetes (ATTD) 2026 Conference, we learned that exciting research is underway. Innovations that apply AI-driven insulin delivery, developments with TZIELD (teplizumab-mzwv) and multiple medications that preserve beta cells at diagnosis.
Here are some top T1D developments to watch for in the upcoming year.
.jpg)
Key Breakthroughs in 2026
Stem Cell-Derived Islet Cell Transplantation (Vertex VX-880)
T1D Strong has reported in the past how Vertex Pharmaceuticals remains a leading biotechnology company in the race to cure type 1 diabetes. Based in Boston’s Seaport District, Massachusetts, with research sites in San Diego, California and Oxford, England, the biotech focuses on T1D cell therapies.
Zimislecel (VX-880) is an investigational, stem cell-derived, islet cell therapy that restores the body’s ability to produce insulin by replacing destroyed pancreatic cells with lab-grown cells that are infused into the liver.
Zimislecel has been granted Fast Track designations from the U.S. Food and Drug Administration. In 2025, clinical trials showed that participants with severe T1D (hypoglycemic unawareness) could achieve insulin independence after 1 year. However, the therapy does require ongoing immunosuppression to prevent islet cell rejection. The zimislecel trial has expanded to additional sites in the U.S., Canada and Europe.
Immunosuppression Therapies Used with Islet Cell Transplantation
Eledon Pharmaceuticals’ tegoprubart is a non-toxic anti-rejection drug designed to protect transplanted (donor-derived) pancreatic islet cells in patients with type 1 diabetes experiencing severe blood sugar swings. Unlike traditional immunosuppressants, it blocks immune cell signals without damaging new cells or causing the harmful side effects of past immunosuppressant therapies.
T1D Strong spoke with Eledon CEO, David-Alexandre Gros, M.D., and Piotr Witkowski, M.D., Ph.D, Director of the Pancreas and Islet Transplant Program at the University of Chicago Medicine’s Transplant Institute, about this groundbreaking, safer path for islet cell transplantation.

The recent trials have shown that tegoprubart enables successful islet transplantation, with some participants being insulin-free for over 12 months.
Gene Therapy (Kriya Therapeutics)
The biotechnology company Kriya Therapeutics is developing KRIYA-839, a one-time intramuscularly injected gene therapy for patients with T1D to produce their own insulin and glucokinase (GCK) to improve glycemic control. It works by using a benign virus to deliver genes that teach those muscles to help regulate blood sugar, just as healthy insulin-producing beta cells in the pancreas do.
The treatment aims to eliminate or significantly reduce the need for daily insulin injections without requiring chronic immunosuppression, with human trials beginning in 2026.
Gene-Edited Hypoimmune Islets (Sana Biotechnology)
Supported by the Breakthrough T1D Fund, Sana Biotechnology is another company working on therapies that do not require immunosuppressant drugs. Sana uses a gene-editing technique called CRISPR-Cas9 to modify donor islet cells so they can evade detection and immune attack. Instead of infusing cells into the liver, the cells are implanted into a patient’s forearm.
This incredible therapy creates “hypoimmune” cells (invisible to the immune system). In the first human study, a T1D individual received a dose of hypoimmune islets without immunosuppressant medication, and the cells appeared to produce insulin. For safety reasons, the participant received only a small dose and continues to receive exogenous insulin; however, additional studies are underway.
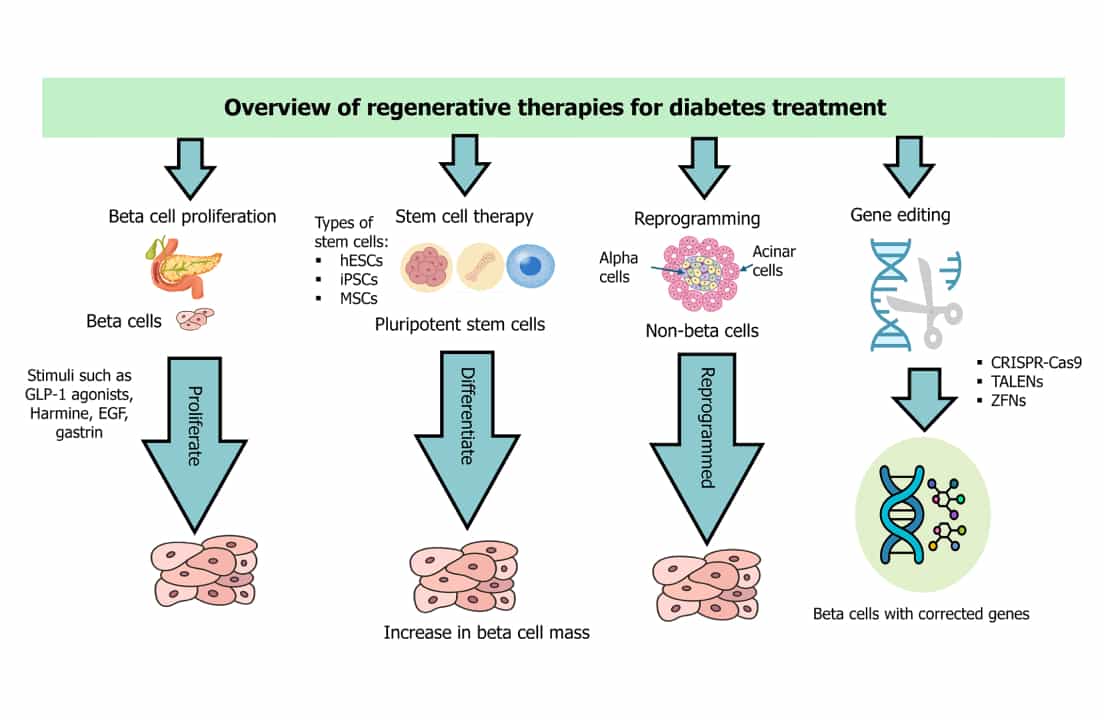
Sana has a new trial (SC451) that uses the same gene-editing strategy with lab-grown, stem-cell-derived insulin-producing cells. The new trial would eliminate the need for long-term immunosuppressant drugs and tackle two key hurdles: cell supply and immune system rejection.
Encapsulation Technology (Sernova Biotherapeutics)
Since type 1 diabetes is an autoimmune condition in which the body attacks insulin-producing cells, research is moving toward the development of encapsulated islets.
Sernova Biotherapeutics’ Cell Pouch™ System is a macro-encapsulation device designed to act as a “bio-hybrid organ.” The chamber enables long-term survival and function of implanted islet-producing cells.
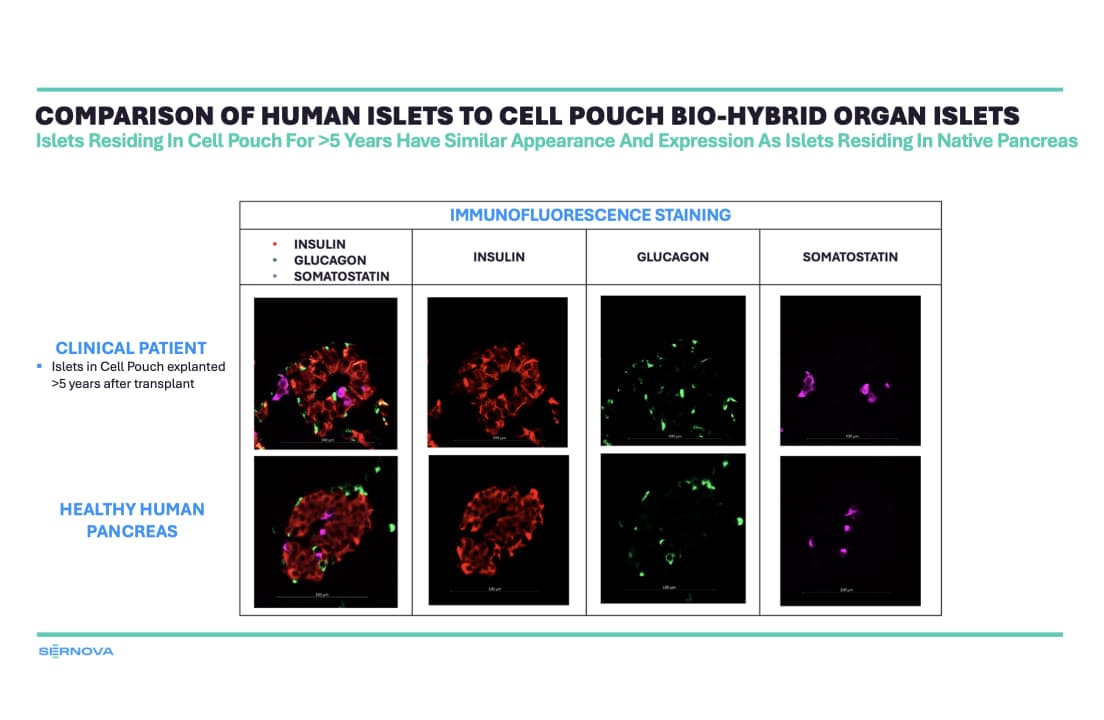
In 2025, T1D Strong spoke with Sernova CEO Jonathan Rigby about Sernova’s efforts toward a T1D functional cure. In 2025, data from Sernova’s Phase 1/2 trials showed that donor islets in their pouches led to insulin independence in 8 of 12 patients. However, this approach did require patients to take immunosuppressants.
The 2026 trial plans to use Evotec’s stem-cell-derived islets instead of donor islets and tegoprubart as an immunosuppressive therapy.
TZIELD (Teplizumab) Expansion
Sanofi’s TZIELD is a prescription immunotherapy that delays the onset of stage 3 T1D in adults and children (8+ years of age) with stage 2 T1D. The drug targets the immune system to slow the destruction of insulin-producing beta cells, delaying insulin dependence for up to two years in some cases.
With a single 2-week course of TZIELD, people had a median of 4 years* before the onset of insulin-dependent type 1 diabetes, compared to 2 years with placebo.
In 2026, Sanofi’s expansion plans include lowering the age range for approval to younger children (age 2) and establishing its use internationally. Ongoing research efforts to explore the medication use in earlier stages (stage 1) in combination with other therapies.
Other 2026 Advancements
AI Automated Insulin Delivery (AIDANET)
In 2026, the Automated Insulin Delivery Adaptive Network (AIDANET) is emerging as a “fully closed-loop” artificial pancreas system. Developed by the University of Virginia (UVA) Center for Diabetes Technology, AIDANET uses artificial intelligence to remove the need for user input regarding meals or exercise.

This AI system learns user patterns to automate insulin delivery. AIDANET integrates a phone app, a continuous glucose monitor (CGM) and an insulin pump.
Recent data from a study of 34 individuals with T1D showed that AIDANET significantly improved glucose control and outperformed standard hybrid closed-loop systems.
First Continuous Ketone Monitor (CKM)
Abbott is developing a dual-action sensor that combines continuous glucose monitoring with continuous ketone monitoring (CKM) to prevent diabetic ketoacidosis (DKA). Based on the small FreeStyle Libre 3 Plus platform, this wearable sensor tracks both glucose and ketone levels in real-time, providing early warnings before ketones reach dangerous levels. No more urine test strips or blood glucose testing required! Potential release expected in 2026.

Screening Advancements
Screening for T1D is gaining popularity in recent years due to the ability to identify high-risk people via antibody testing, allowing them to delay disease onset with therapies like Tzield and avoid life-threatening diabetic ketoacidosis (DKA).
T1D Scout, the first saliva-based at-home test kit, recently highlighted the importance at the ATTD conference, showing how early screening may improve long-term health monitoring, preserve insulin production and save lives by preventing life-threatening conditions like DKA.

Teamwork Makes the Dream Work
While all the T1D advancements are encouraging, we know that discoveries overlap. The need for a collaborative approach in type 1 diabetes research has never been as important as it is in 2026.
The current landscape is promising as the scientific community recognizes that breakthroughs in cell therapies, immunotherapy, and technology are most effective when developed together.


.webp)





.webp)

.jpeg)




.jpg)






.jpg)



%20(1).jpg)





.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)

.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)


.jpg)

.jpg)



.jpg)
.jpg)



.jpg)



.jpg)

.jpg)




.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)


.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)

.jpg)




.jpg)
.jpg)
.jpg)
.jpg)







.webp)