T1D Guide
T1D Strong News
Personal Stories
Resources
T1D Misdiagnosis
T1D Early Detection
Research/Clinical Trials
From Screening Her Children to Starting Tzield: Chris Dunn’s Type 1 Diabetes Journey
Chris Dunn is no stranger to type 1 diabetes (T1D). Her son Nolan was diagnosed at two years old, and her daughter, Patsy, followed at age four. Having two young children with type 1 is hard for many to imagine. It’s the reason many parents push the idea of screening aside, but Dunn said it was the best decision she’s ever made. Through early detection, she discovered she carries T1D autoantibodies and has started Tzield, a disease-modifying therapy, to delay onset.
.jpg)
The Impact of T1D on a Family
In 2006, Chris Dunn and her husband Bob received a shock when their two-year-old son, Nolan, was diagnosed with type 1 diabetes (T1D). Besides a cousin and Bob’s uncle, they had no family history of T1D. A critical issue with type 1 toddlers and infants is that they can’t articulate how they’re feeling.
“I just have a vivid memory of him, standing by the refrigerator crying for drinks.” Fortunately, Dunn had a background in sports medicine and recognized the symptoms as they became more frequent. She called her pediatrician, asking to test for type 1 diabetes, but there was no appointment until Monday. Dunn conveyed the urgency to see someone right away.
At the doctor’s office, Nolan’s blood sugar tested so high it didn’t even register on the meter, so it was an immediate transport to the children’s hospital.
“He was in DKA (diabetic ketoacidosis). We were in the hospital for a few days,” said Dunn. “I’ve got this very vivid memory of looking at this tiny little guy in that big bed and thinking, ‘How do I send this kid to college?”
Thanks to the many technological breakthroughs over the years, Nolan is a senior at the University of Nebraska-Lincoln. But at the time of his diagnosis, it was five shots and 20 finger pokes a day, midnight and three a.m. checks every night. This was before the age of CGMs (continuous glucose monitors) and smartphones.
“I’d turn on all the lights and play this handheld Yahtzee game to stay awake to re-check him. I still have the game on my bedside table, just as a symbol of everything we lived through,” Dunn said. “I think I still might have a little PTSD just from dragging that poor little guy out from where he would try to hide behind the recliner because he didn’t want to get poked anymore.”
A Second Diagnosis
Six years later, in 2012, Dunn’s daughter Patsy was diagnosed at age four.
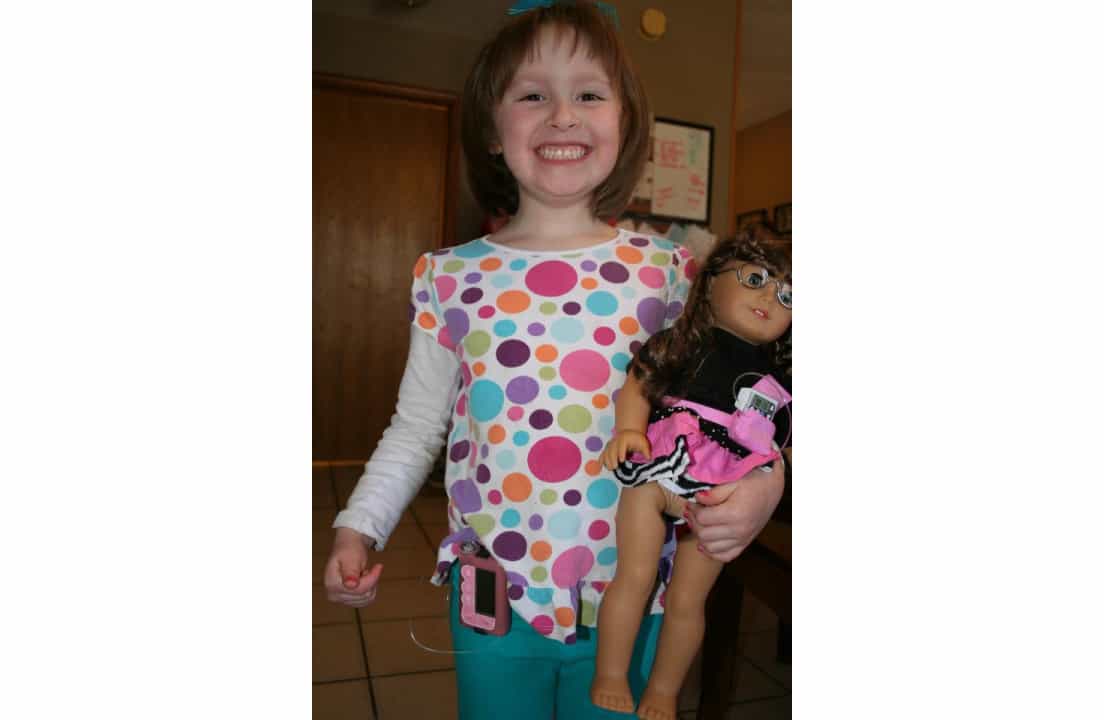
A year earlier, they tried to get her screened through TrialNet, with her daughter Ellie, but Patsy was so little they couldn’t get enough blood, and after poking her several times, Dunn stopped them.
“We had the heightened awareness of the signs and symptoms, so we caught her very early, and she did not have to be hospitalized; she was not in DKA, and what’s interesting, to this day, Patsy has an easier time managing her blood sugar. She has a better A1c, better time-in-range (TIR), because she was never in DKA.”
Studies with the National Institutes of Health (NIH) confirm that children and youth diagnosed with type 1 diabetes without diabetic ketoacidosis achieve better long-term glycemic control, including lower blood sugar levels and higher time-in-range (TIR), compared to those who present with DKA at the time of diagnosis.
Dunn went on to say that she thinks everyone’s experience with T1D is just a little bit different. “They react to foods differently, and exercise, so it’s just a matter of getting to know each of them and treating them very uniquely for their own good.”
Four Children Under Ten: Two with T1D
With two T1Ds under eight, and two other children under age 10, it was challenging to say the least. “You just feel like you’re living in a hole, trying to keep your child alive 24 hours a day,” said Dunn.

“When we went to that first Breakthrough T1D (Formerly JDRF) Walk, I don’t think there’s anything more therapeutic. There was now this whole community of people that I could be a part of. I felt empowered to change my son’s future. And to this day, I can see where the research is going. Thankfully, we’ve got technology now that makes life much easier and safer.”
Today, both Patsy (17) and Nolan (22) are on the Beta Bionics iLet insulin pump and Dexcom CGM.
Knowledge is Power: Reasons to Screen
Identifying the disease before symptoms appear in its early stages prepares families to manage insulin dependence. Aside from preventing the life-threatening condition of DKA, we now know that you can safely delay the progression of T1D for several years through clinical trials.
“Not knowing isn’t going to make it not happen, and in my opinion, knowledge is power. The data is very clear that screening helps prevent DKA at diagnosis. And now we have an FDA-approved therapy that could delay the onset of stage 3 T1D. Why would you not want to know that so that you could potentially have that opportunity?” Dunn asks.
Any day you can stay off insulin therapy and all of the risks, it’s worth it to me – Chris Dunn.
Other positive aspects of screening include time to prepare both financially and mentally.
“We don’t know how long a delay you’re going to get,” said Dunn. “We know the average is two to three years, but for some people, it’s been much longer. There have been people who received their Tzield infusion for a decade or more because they were part of the original clinical trial.”
Stages of Type 1 Diabetes
Knowing that type 1 diabetes progresses through four stages is crucial because it enables proactive monitoring and allows families to prepare rather than react. It also opens the door to dozens of ongoing clinical trials.
.jpg)
“Just look at where we’re at in our progress towards curative therapy, specifically with manufactured islet replacement therapies that are coming down the pipeline,” said Dunn. “If you can get a delay and then these therapies are on the horizon, why would you not want to try to delay it so that you would have an option of one of these other therapies rather than insulin therapy?”
Dunn’s Screening Experience through ASK
In 2011, Dunn screened through TrialNet and tested negative for all five autoantibodies associated with T1D. “My understanding at the time was if you screen negative as an adult, you’re not going to seroconvert—you’re good.”
Then, in the spring of 2025, the Dunn family completed follow-up screening through the Autoimmunity Screening for Kids (ASK) program at the Barbara Davis Center (BDC) for Diabetes at the University of Colorado. Dunn’s daughter, Mary Kate, was due to be re-screened, and on impulse Dunn ordered the test kit for herself.
She knew that more than 50% of diagnoses occur in adults, so to promote the importance of screening people of all ages, she decided to test herself, too.
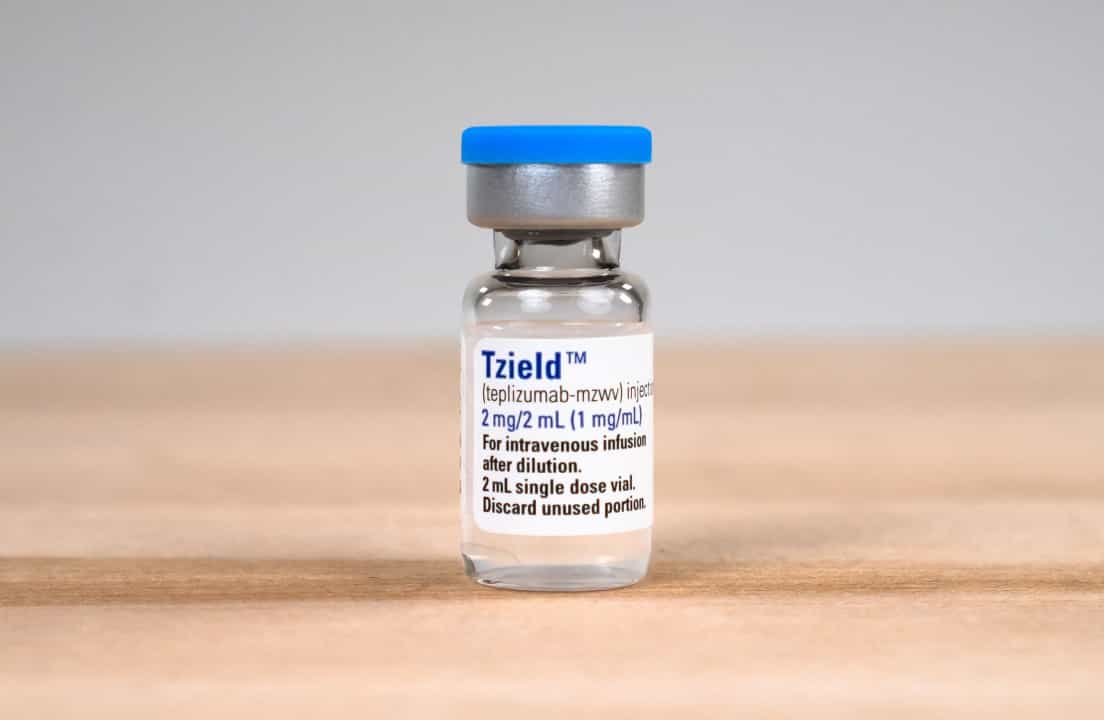
A few weeks later, Dunn received a call from ASK informing her that she had tested positive for four autoantibodies. “I thought, well, that changes things.” They asked whether she was familiar with Tzield (teplizumab-mzwv), the FDA-approved medication used to delay the onset of stage 3 type 1 diabetes in adults and children (ages 8 and older).
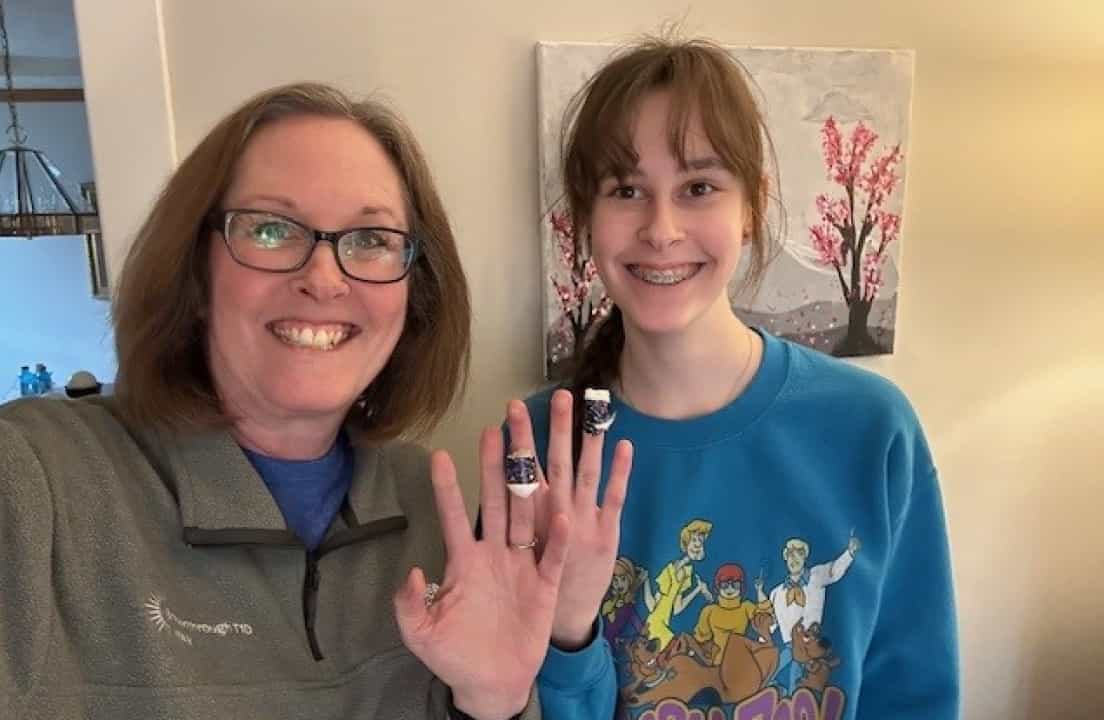
“I said, yep, I’m actually very familiar with Tzield. In fact, the day Tzield got approved by the FDA, we celebrated with some T1D families locally, toasting with champagne to the fact that we had the first-ever FDA-approved disease-modifying therapy for type 1 diabetes, never imagining that I would be the recipient of that therapy just a few years later.”
The first step after receiving the call was to confirm her autoantibody status and to receive education on T1D. “I honestly think I rolled my eyes. I’ve been doing this for 20 years. But I didn’t know diddly squat about stage 2.”
“I knew about stage 3 and stage 4 T1D, but I learned that kids typically progress through stage 2 very quickly, where adults can live in stage 2 for decades,” said Dunn.

“Tzield is most effective right at the end of stage 2. So, even though we knew I was in stage 2 when I left my appointment that day, we had to wait for my C-peptide results to come back to see how much beta cell function was actually left.”
Based on those results, her physician at the Early T1D BDC Clinic told Dunn there was quite a bit of beta cell damage and that she should proceed with Tzield sooner rather than later, or they could miss the window of opportunity.
What Tzield Infusions Look Like
Dunn’s first infusion started on July 28, 2025, and each day was different. Her first day was the longest, because she had to undergo the outpatient surgical procedure to insert the PICC line.
This was followed by lab work to ensure it was safe to proceed with the infusion, and pre-medication to minimize side effects. “The infusion itself was pretty short. I think maybe 30 minutes,” said Dunn. “Afterwards, you have to stay for another hour to monitor vitals and make sure it’s safe for you to leave. So the first day, we were there for about seven to eight hours.”
After that, the infusions, which last 14 days, were much shorter, with some sessions as short as an hour and a half.
Tzield Common Side Effects include:
- Flu-like symptoms (fever, nausea, fatigue, muscle/joint pain)
- Rash or allergic reactions
- Headache
- Lymphopenia/Leukopenia: A decrease in white blood cells
- Stomach pain (diarrhea, vomiting)
Dunn explained that over the past several years, the technicians administering the infusions have learned to start with a really low dose, and then, for the first five days, your dose basically doubles what it was the previous day.
“So day one is very low. Then day two doubles what you had on day one,” Dunn said. “Day three doubles what you had on day two, and it continues until you get to day five. Days five and six were the most challenging. Those were the first days when you received the full dose of the medication. So, it felt similar to influenza, where the body aches with a little headache and nausea.”
“There’s also a rash that’s very common with Tzield, but what’s interesting is it seems to manifest differently from person to person, and mine was on the palms of my hands and the soles of my feet. But again, the medication helps to alleviate symptoms until it clears up completely, and then by day seven, the symptoms were on their way out,” Dunn said. “They just continued to get better as we went.”
Moving Forward After Initial Infusions
Since her stage 2 diagnosis, Dunn has experienced some hypoglycemic episodes (without insulin therapy). The doctor explained that in stage 2, because you do have beta cell damage, the cells try to work and secrete insulin, but they can actually overshoot.
For now, Dunn manages her condition more like type 2 diabetes (T2D). Certain foods (bananas, apples, soft pretzels) seem to spike her blood sugar. “A lot of it is just being smart. Lower carb options are gonna be better for my blood sugar, and I know that it’s better long-term for my body not to have elevated blood sugar.”
“For me, it’s helpful to see exactly how my blood sugar reacts to specific foods, so that I can control my blood sugar and keep it more in range by what I’m choosing to eat.”
Another interesting aspect Dunn experienced while managing her T1D through diet and CGM was that her extreme hip and knee pain went away. “I really think it was systemic inflammation, because my blood sugar was elevated. For me, that’s another reason to continue managing it more tightly, because it has dramatically increased my quality of life.”
Different T1D Genotypes & Tzield Outcomes
Just as each individual with type 1 manages their diabetes differently and responds to food and exercise in their own way, each person responds to Tzield differently. “I know of people who receive this therapy, and six months later they’re on insulin, but then I know of other people who were in the original clinical trial, and it’s been 11 years, and they’re still not taking insulin.”
Additionally, during her Tzield therapy, Dunn spoke with a physician who believes there are different genotypes of type 1, with some being good responders to Tzield and others not.
“He said, the ultimate goal is that eventually we’ll have universal screening for type 1 diabetes, so if someone tests positive for autoantibodies, we determine which genotype they have. Then hopefully, we’ll have multiple disease-modifying therapies (DMTs) and a menu of options for the best outcome.”
Tzield is currently the only FDA-approved therapy, but multiple DMT options are expected to be readily available in the future.
Progress on the T1D Horizon
Dunn remains hopeful that she’ll continue to respond positively to the Tzield therapy. When asked what she’s most excited about on the T1D path to possible cures, she said, “The work with manufactured islet cell transplants is incredible. This curative therapy is coming, and it’s not a matter of if we’re going to cure type 1 diabetes; it’s a matter of how fast we can do it.”
For more information, here is the link to Breakthrough T1D’s early detection page with screening options.


.webp)





.webp)







.jpeg)


%20(1).jpg)




.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)


.jpg)

.jpg)


.jpg)
.jpg)

.jpg)

.jpg)
.jpg)


.jpg)
.jpg)
.jpg)
.jpg)


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)


.jpg)
.jpg)
.jpg)

.jpg)
.jpg)


.jpg)

.jpg)
.jpg)

.jpg)















.webp)