T1D Guide
T1D Strong News
Personal Stories
Resources
T1D Misdiagnosis
T1D Early Detection
Research/Clinical Trials
Tzield Just Approved for Ages 1 and Up in Stage 2 T1D
In a significant new development, the U.S. Food and Drug Administration (FDA) has approved Sanofi’s Tzield (teplizumab-mzwv) for toddlers with stage 2 type 1 diabetes (T1D), making it the first disease-modifying therapy available for children aged one and older at this stage.

As screening for type 1 diabetes becomes more widespread, this milestone offers new hope for families seeking to delay the onset of the condition in other children. Previously approved for children ages eight and older, this expanded FDA approval is supported by evidence from the Phase 4 PETITE-T1D study.
Having a toddler with T1D, one who can’t articulate what they’re feeling, can be a frightening experience. As most parents with one child know, T1D requires 24/7 vigilance, constant care to keep blood sugar levels from dropping too low (hypoglycemia) or rising too high (hyperglycemia).

Both directions can be life-threatening with side effects like diabetic ketoacidosis (DKA) if levels remain high for too long, and unconsciousness or coma if levels slip too low.
Besides the fact that growing toddlers can’t communicate their symptoms, it’s very difficult to manage the complexity of a young child’s growth spurts, activity level and food intake.
Despite advances in continuous glucose monitors (CGMs), constant monitoring can be exhausting for parents. It may also contribute to anxiety and burnout, making the ability to delay the disease with a disease-modifying therapy a remarkable breakthrough.
What is Tzield
The disease-modifying therapy, Tzield, acts as a ‘pause button’, halting the development of type 1 diabetes. We now know that T1D develops in stages and that screening can detect the autoantibodies that indicate a type 1 diagnosis.
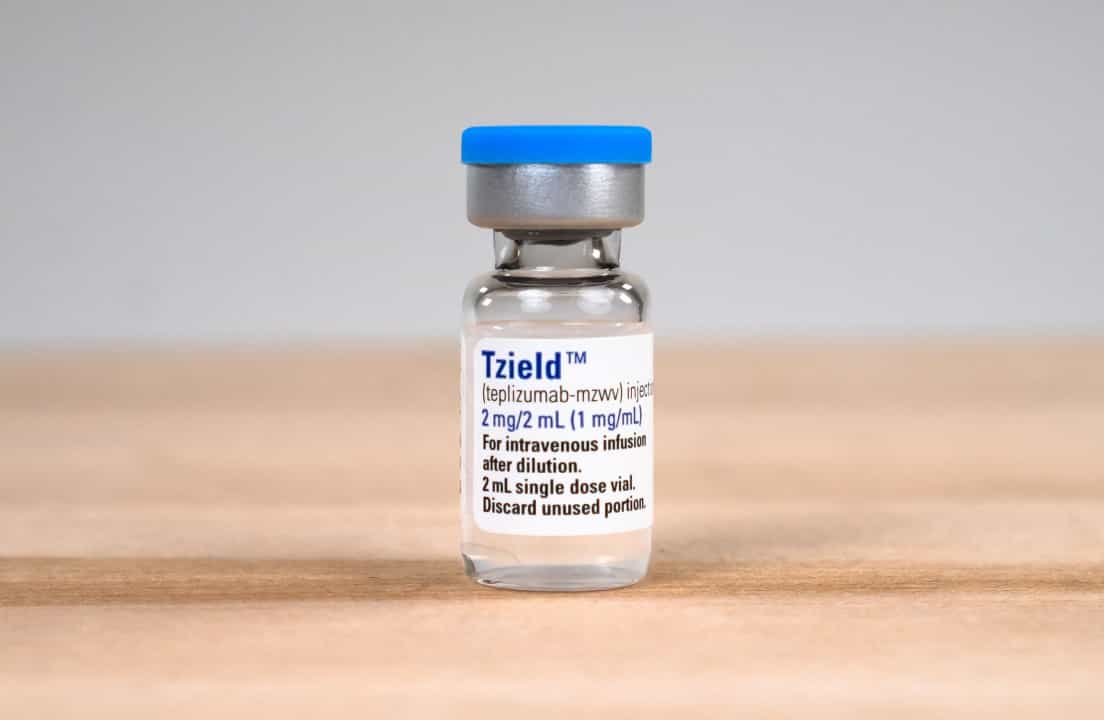
Here’s a Tzield Breakdown:
How it Works
Tzield works by targeting the immune system to slow the destruction of insulin-producing cells. As Tzield slows down the immune system’s attack on the pancreas, it allows the beta cells to continue making insulin for a longer period.
The treatment is given as an IV infusion (an injection into the vein) once daily for 14 consecutive days.
Who is it for?
The therapy is available for adults and children with stage 2 type 1 diabetes. Testing positive for at least two types of diabetes-related autoantibodies and having abnormal blood glucose levels, but not yet experiencing the need to take daily insulin.
Key Benefits
Studies showed it delayed the need for insulin by an average of roughly two years (some saw longer delays).

Side Effects
There are reported side effects, ranging from mild rashes, headaches and flu-like symptoms to more serious effects such as decreased white blood cell counts. But first-hand reports say these side effects diminish as the treatment ensues.
Benefits of Tzield
Most importantly, Tzield gives families time to weigh their options and prepare for a type 1 diabetes diagnosis, which can be financially expensive and emotionally trying.
To avoid the immediate need for daily insulin management, avoid the life-threatening risks and plan for the necessary lifestyle changes that come with type 1 diabetes.
Consequently, this approved therapy is only getting better, with scientists working hard to extend the offset duration.
For more information on screening:
TrialNet: Offers free risk screening for relatives of people with T1D and for the general population.
ASK (Autoimmunity Screening for Kids): A free program for children ages 2–17 that can be done at home or at local labs.
Pediatrician/Primary Care Office: Ask your pediatrician to order an islet autoantibody panel.
T1D Scout: This is a fully remote, at-home, saliva-based screening program designed to detect early-stage T1D risk in children 0–18 and adults, without initial blood draws.

Specialized Screening Initiatives: Programs such as the PLEDGE study (through Sanford Health) offer screenings during routine appointments.


.webp)





.webp)





%20(1).jpg)



.jpeg)


.jpg)

.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)



.jpg)

.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)



.jpg)
.jpg)



.jpg)



.jpg)

.jpg)




.jpg)
.jpg)
.jpg)


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)

.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)




.jpg)
.jpg)
.jpg)


.jpg)
.jpg)








.webp)